The complex structural chemistry brought about by the different compositions of layered metal oxides (NaxTMO2, TM=transition metal) plays a decisive role in the layered stacking structure, sodium ion conductivity and redox activity, opening up the study of functional materials New way. NaxTMO2 mainly includes two structures, O3 and P2, and the P2 structure has a faster Na+ diffusion rate due to the open triangular prism diffusion channel. However, the initial charging capacity of the P2-type structure is low (<80 mAh g-1), and the average voltage is less than 3.2 V. When charging to a higher voltage, it will undergo a phase change from P2 to O2 or OP4/"Z" resulting in cycle stability. decline.
Sodium content is a key factor affecting the structural stability of P2. During the charging process, the P to O phase transition will occur as the TMO2 layer slips due to the extraction of sodium. If the sodium content in the P2 structure is sufficient during charging, the P2 structure can also remain stable during charging and discharging. At the same time, the higher sodium content in the P2 structure makes the average valence of TM lower, enabling higher sodium storage capacity at lower voltages. Therefore, it is important to systematically investigate the effect of sodium ion content in the P2 phase layered oxide on the structure and properties of the material.
Under the guidance of Hu Yongsheng, researcher of the Institute of Physics, and Marnix Wagemaker, a researcher at the Institute of Physics, Dr. Chenglong Zhao of the E01 group of the Clean Energy Key Laboratory of the Institute of Physics of the Chinese Academy of Sciences/Beijing National Research Center for Condensed Matter Physics studied the P2 layered oxidation Potentially the highest sodium ion content in the substance, and it was observed that high sodium content can improve the stability of the structure, and at the same time can promote the oxidation of low-valent cations to high-valence states. For the sodium ion P2 phase material, it is very important to realize the transition from Ni2+ to Ni4+ at a voltage of 4.0 V. Under normal circumstances, the transition from Ni2+ to Ni4+ will occur above about 4.2 V. Such high voltage is usually accompanied by structural evolution, electrolyte decomposition, and oxygen loss due to oxygen ion oxidation, etc., causing the material to suffer performance degradation during cycling . Introducing a high sodium content in the P2 phase structure will reduce the average valence state of the transition metal ions in the structure, which will promote the transition of the transition metal with the lowest oxidation state to its high valence state and achieve higher capacity. During the sodium removal process, a large amount of sodium ions in the P2 phase structure can greatly improve the stability of the structure.
For P2 phase cathode materials with low sodium content, when charged and when the sodium ion content in the structure is less than about 1/3, the original P2 phase structure will be transformed into O2 phase or OP4/"Z" phase. When the same sodium ion is extracted, the high sodium content can leave more sodium in the structure, weaken the repulsion between adjacent transition metal layers, and inhibit the transition metal layer slip. For P2-Na45/54Li4/54Ni16/54Mn34/54O2 materials with high sodium content, there is no transition from P to O phase during charging; the newly formed P2 phase has the same structure as the original P2 phase, and has more The large sodium ion layer spacing will promote the diffusion of sodium ions to a certain extent and achieve higher reversibility. Studies have shown that within a 2.0-4.0 V electrochemical window, the material can achieve a multi-electron transfer reaction from Ni2+ to Ni4+, showing a high reversible capacity of ~100 mAh g-1, and stable cycling. Compared with the traditional low sodium content P2 phase materials, the high sodium content P2 phase materials provide new insights into the electronic structure and chemical structure for the development of new electrode materials to a certain extent.
The research results are published in the Journal of the American Chemical Society under the title of Revealing High Na-Content P2-Type Layered Oxides for Advanced Sodium-Ion Cathodes. Relevant work was supported by the National Natural Science Foundation of China, the Strategic Leading Science and Technology Project of the Chinese Academy of Sciences, the Beijing Municipal Science and Technology Commission and the Beijing Natural Science Foundation-Haidian Original Innovation Joint Fund, and the National Public Education Fund Committee.
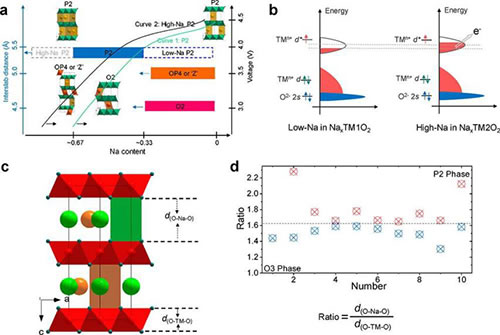
Figure 1. (a) Structure evolution mechanism of P2 oxide with high sodium content during sodium removal; (b) Electronic structure of P2 oxide with low/high sodium content; (c) Crystal structure of P2 oxide; (d) dO -The ratio of Na-O and dO-TM-O in P2 and O3 compounds
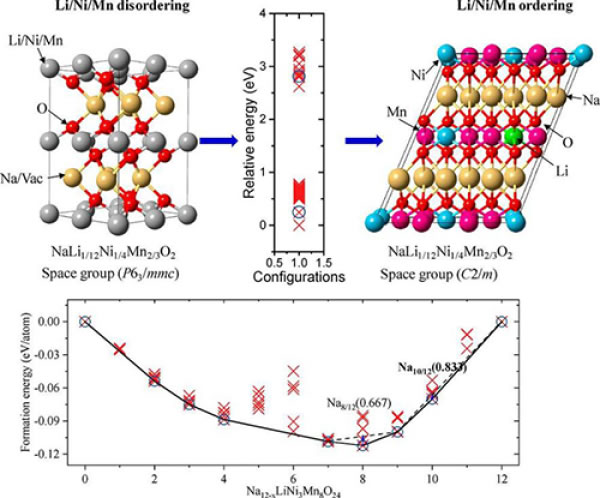
Figure 2. (a) XRD spectra of Na45/54Li4/54Ni16/54Mn34/54O2; (b) HAADF and ABF-STEM in [010] direction; (c) EELS spectra of Na, Mn, Ni and O
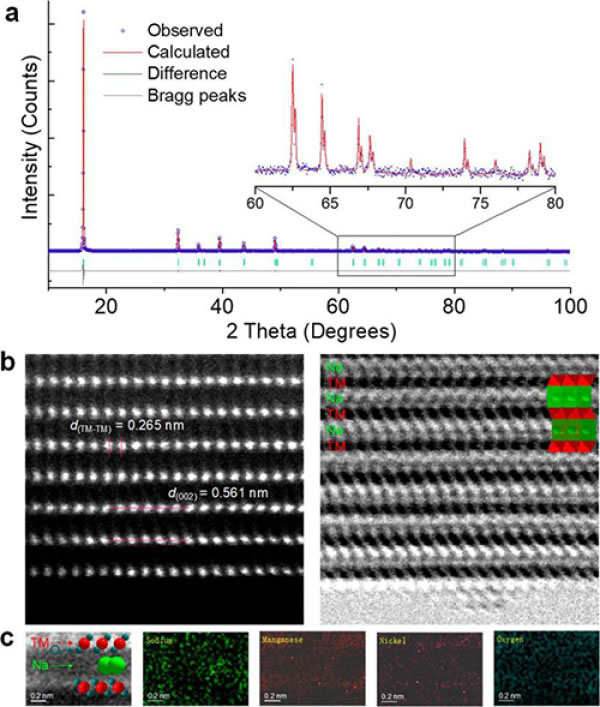
Figure 3. (a) Li/Ni/Mn arrangement in Na12-xLiNi3Mn8O24; (b) phase stability in Na12-xLiNi3Mn8O24-LiNi3Mn8O24
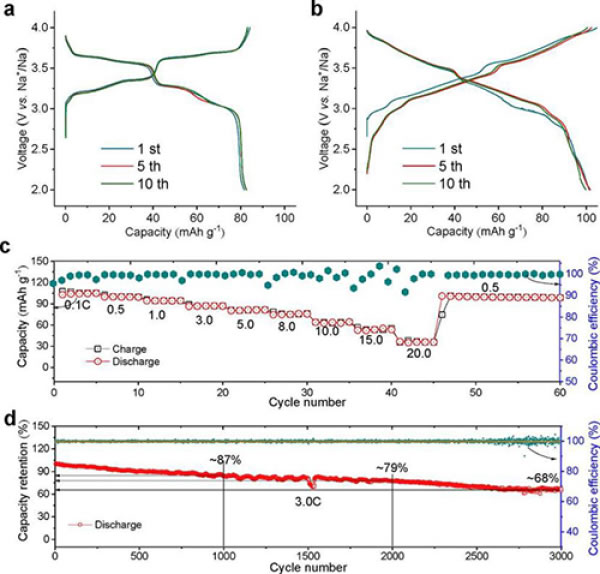
Figure 4. (a) Charge and discharge curves of Na2/3Ni1/3Mn2/3O2 at 0.15C (~22 mA g-1), 2-4 V: (a) Na2/3Ni1/3Mn2/3O2, (b) Na45/ 54Li4/54Ni16/54Mn34/54O2; (c) rate performance of Na45/54Li4/54Ni16/54Mn34/54O2; (d) discharge capacity retention rate of Na45/54Li4/54Ni16/54Mn34/54O2
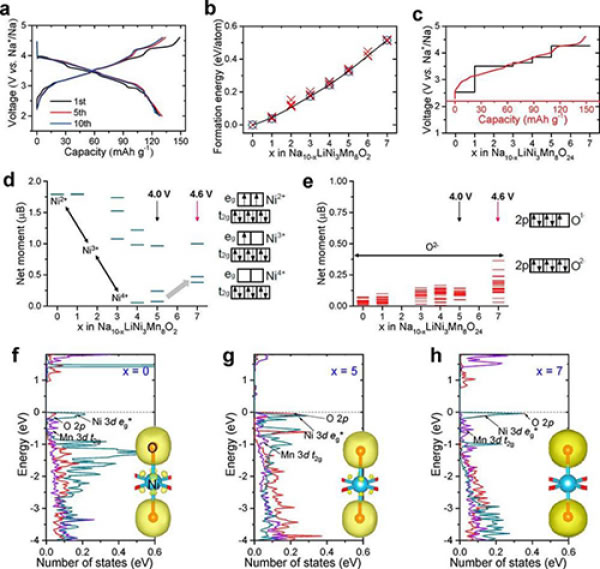
Figure 5. (a) Na45/54Li4/54Ni16/54Mn34/54O2 charge-discharge curve at 2-4.6 V, 0.15 C; (bc) Na10-xLiNi3Mn8O24 (0≤x≤7) formation energy of different sodium removal structures; ( de) Magnetization and oxidation states of Ni and O in the mesophase of Na10LiNi3Mn8O24 to Na3LiNi3Mn8O24 during sodium removal; (fh) Electronic structure evolution of Ni 3d t2g, Mn 3d t2g and O 2p at x=0, x=5 and x=7
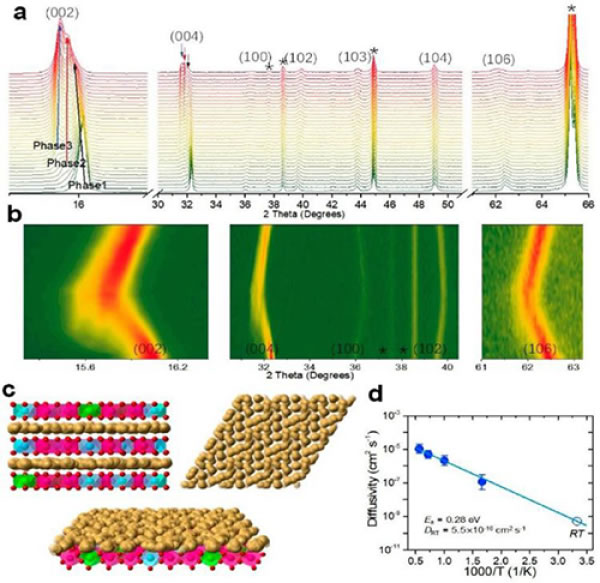
Figure 6. (a) 0.05 C, in-situ XRD spectrum charged to 4.6 V; (b) 0.5 C, 2-4 V in-situ XRD spectrum; (c) Na+ in P2 structure with high sodium content Migration path; (d) Arrhenius point of diffusion coefficient obtained by molecular dynamics simulation
106Cm Pvc Wallpaper,Pvc White Home Decor Wallpaper,Wall Home Decoration,Bedroom Living Room Tv Sticker
JIANGSU ARTSTYLE DECORATION MATERIALS CO..LTD , https://www.jsadpvcwallpaper.com